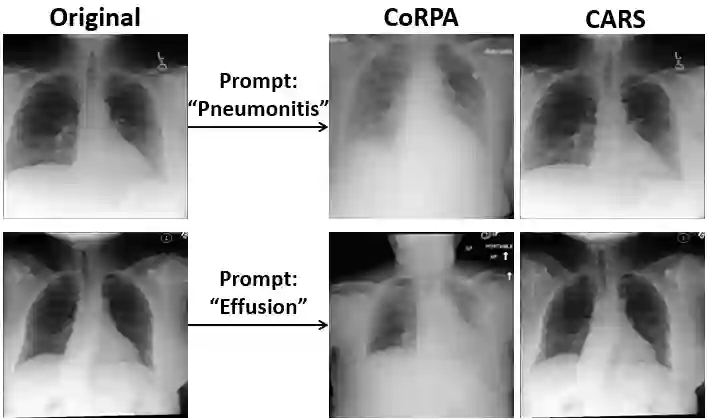
The clinical deployment of AI diagnostic models demands more than benchmark accuracy - it demands robustness across the full spectrum of disease presentations. However, publicly available chest radiographic datasets systematically underrepresent critical clinical feature combinations, leaving models under-trained precisely where clinical stakes are highest. We present CARS, a clinically aware and anatomically grounded framework that addresses this gap through principled synthetic image generation. CARS applies targeted perturbations to clinical feature vectors, enabling controlled insertion and deletion of pathological findings while explicitly preserving anatomical structure. We evaluate CARS across seven backbone architectures by fine-tuning models on synthetic subsets and testing on a held-out MIMIC-CXR benchmark. Compared to prior feature perturbation approaches, fine-tuning on CARS-generated images consistently improves precision-recall performance, reduces predictive uncertainty, and improves model calibration. Structural and semantic analyses demonstrate high anatomical fidelity, strong feature alignment, and low semantic uncertainty. Independent evaluation by two expert radiologists further confirms realism and clinical agreement. As the field moves toward regulated clinical AI, CARS demonstrates that anatomically faithful synthetic data generation for better feature space coverage is a viable and effective strategy for improving both the performance and trustworthiness of chest X-ray classification systems - without compromising clinical integrity.
翻译:AI诊断模型的临床部署不仅需要基准精度,更要求在全谱疾病表现中具备鲁棒性。然而,公开的胸部放射影像数据集系统性地缺失关键临床特征组合,导致模型在临床风险最高的场景下训练不足。我们提出CARS——一个临床感知且解剖学基础的框架,通过基于原理的合成图像生成来弥补这一缺陷。CARS对临床特征向量实施定向扰动,在明确保持解剖结构的同时,实现病理征象的可控插入与删除。我们通过在合成子集上微调模型并在保留的MIMIC-CXR基准上进行测试,评估了七种骨干架构的性能。与先前的特征扰动方法相比,基于CARS生成图像进行微调能持续提升查准率-查全率性能,降低预测不确定性,并改善模型校准度。结构与语义分析显示其具有高解剖保真度、强特征对齐性和低语义不确定性。两位放射学专家的独立评估进一步证实了其真实性与临床一致性。随着领域向受监管的临床AI迈进,CARS证明:通过解剖学保真的合成数据生成以改善特征空间覆盖,是提升胸部X光分类系统性能与可信度的可行且有效的策略——且不损害临床完整性。