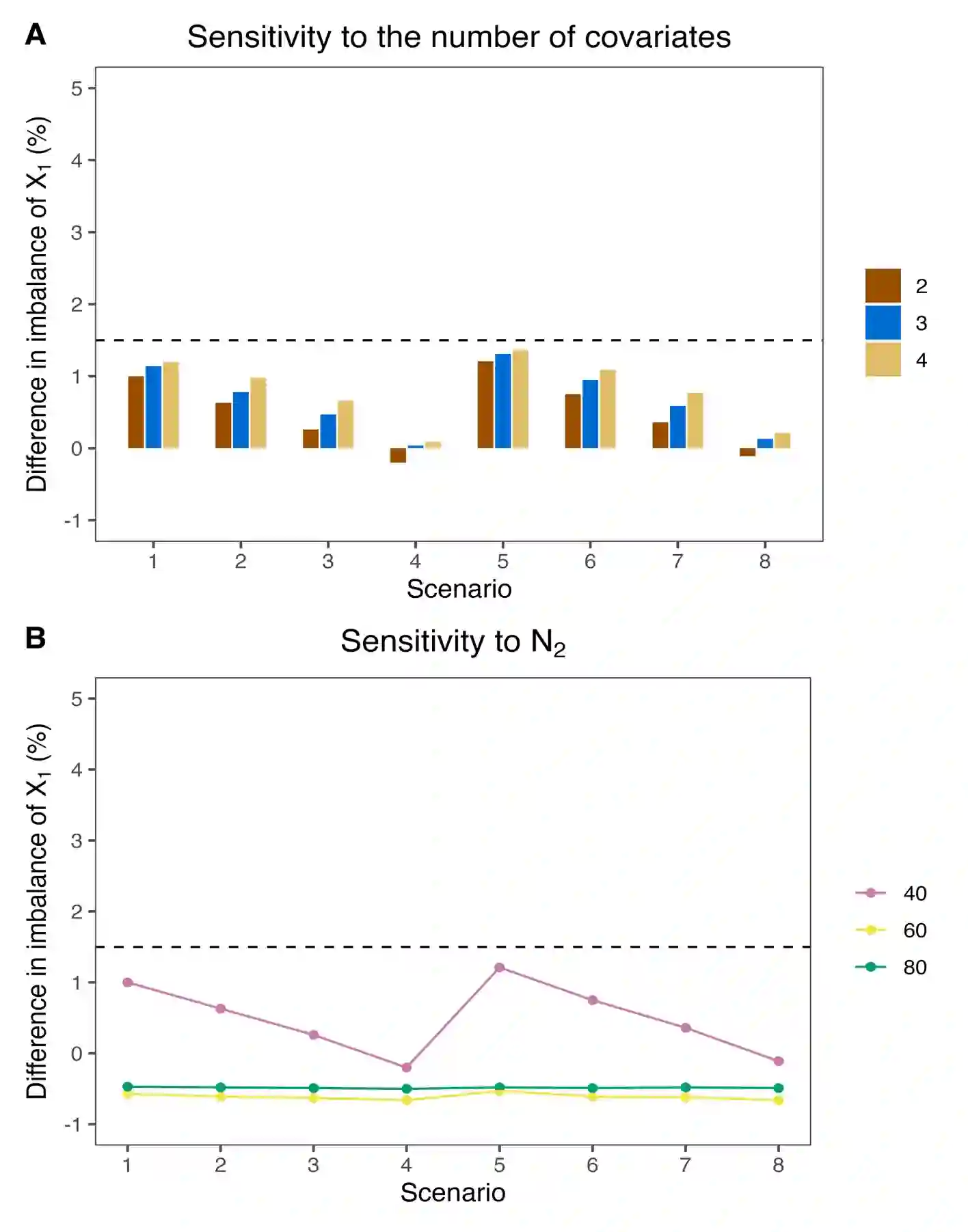
One common approach for dose optimization is a two-stage design, which initially conducts dose escalation to identify the maximum tolerated dose (MTD), followed by a randomization stage where patients are assigned to two or more doses to further assess and compare their risk-benefit profiles to identify the optimal dose. A limitation of this approach is its requirement for a relatively large sample size. To address this challenge, we propose a seamless two-stage design, BARD (Backfill and Adaptive Randomization for Dose Optimization), which incorporates two key features to reduce sample size and shorten trial duration. The first feature is the integration of backfilling into the stage 1 dose escalation, enhancing patient enrollment and data generation without prolonging the trial. The second feature involves seamlessly combining patients treated in stage 1 with those in stage 2, enabled by covariate-adaptive randomization, to inform the optimal dose and thereby reduce the sample size. Our simulation study demonstrates that BARD reduces the sample size, improves the accuracy of identifying the optimal dose, and maintains covariate balance in randomization, allowing for unbiased comparisons between doses. BARD designs offer an efficient solution to meet the dose optimization requirements set by Project Optimus, with software freely available at www.trialdesign.org.
翻译:剂量优化的一种常见方法是两阶段设计,即首先进行剂量递增以确定最大耐受剂量(MTD),随后进入随机化阶段,将患者分配至两个或多个剂量组,以进一步评估和比较其风险-获益特征,从而确定最佳剂量。该方法的一个局限在于需要相对较大的样本量。为应对这一挑战,我们提出了一种无缝两阶段设计——BARD(用于剂量优化的回填与自适应随机化),该设计整合了两个关键特性以降低样本量并缩短试验周期。第一个特性是将回填整合到第一阶段剂量递增中,在不延长试验时间的前提下提高患者入组率和数据生成效率。第二个特性是通过协变量自适应随机化,无缝整合第一阶段与第二阶段接受治疗的患者,从而为确定最佳剂量提供信息并减少样本量。我们的模拟研究表明,BARD能够减少样本量,提高识别最佳剂量的准确性,并在随机化过程中保持协变量平衡,从而实现剂量间的无偏比较。BARD设计为满足"Optimus项目"设定的剂量优化要求提供了一种高效解决方案,相关软件可在 www.trialdesign.org 免费获取。