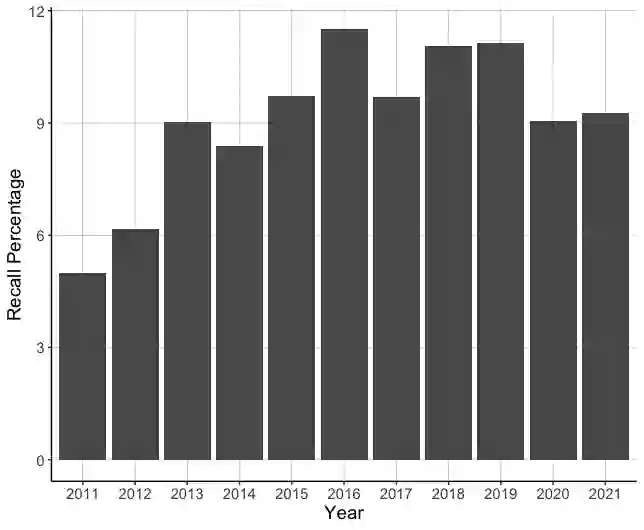
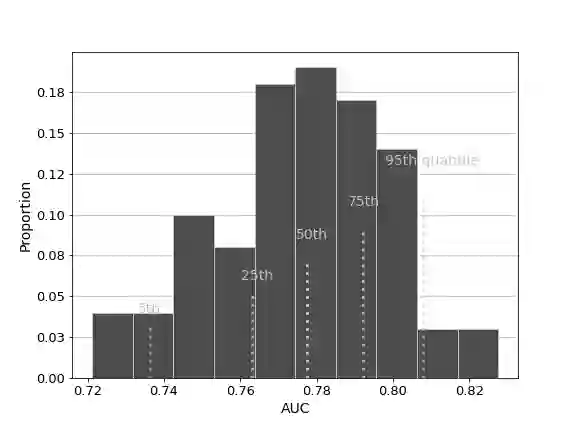
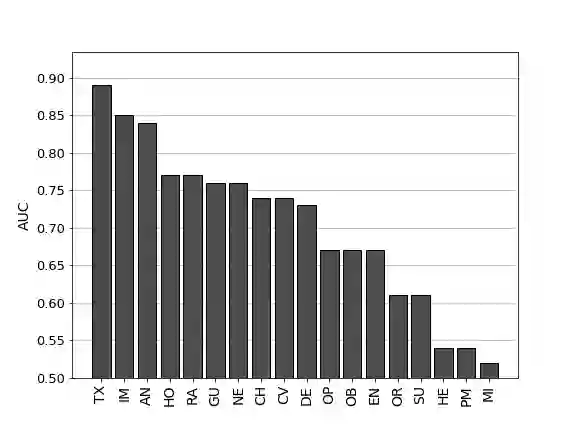
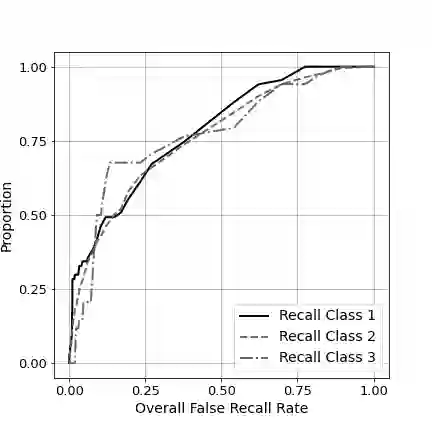
The United States Food and Drug Administration's (FDA's) 510(k) pathway allows manufacturers to gain medical device approval by demonstrating substantial equivalence to a legally marketed device. However, the inherent ambiguity of this regulatory procedure has been associated with high recall among many devices cleared through this pathway, raising significant safety concerns. In this paper, we develop a combined human-algorithm approach to assist the FDA in improving its 510(k) medical device clearance process by reducing recall risk and regulatory workload. We first develop machine learning methods to estimate the risk of recall of 510(k) medical devices based on the information available at the time of submission. We then propose a data-driven clearance policy that recommends acceptance, rejection, or deferral to FDA's committees for in-depth evaluation. We conduct an empirical study using a unique dataset of over 31,000 submissions that we assembled based on data sources from the FDA and Centers for Medicare and Medicaid Service (CMS). Compared to the FDA's current practice, which has a recall rate of 10.3% and a normalized workload measure of 100%, a conservative evaluation of our policy shows a 32.9% improvement in the recall rate and a 40.5% reduction in the workload. Our analyses further suggest annual cost savings of approximately $1.7 billion for the healthcare system driven by avoided replacement costs, which is equivalent to 1.1% of the entire United States annual medical device expenditure. Our findings highlight the value of a holistic and data-driven approach to improve the FDA's current 510(k) pathway.
翻译:美国食品药品监督管理局(FDA)的510(k)审批途径允许制造商通过证明其产品与已合法上市器械具有实质性等同性而获得医疗器械批准。然而,该监管程序固有的模糊性导致通过此途径获批的众多器械召回率居高不下,引发了严重的安全隐忧。本文提出一种人机协同方法,旨在协助FDA改进其510(k)医疗器械审批流程,以降低召回风险并减轻监管负担。我们首先开发了机器学习方法,基于器械提交时可获取的信息来评估510(k)医疗器械的召回风险。随后提出一种数据驱动的审批策略,可向FDA委员会建议接受、拒绝或转交深入评估。我们通过对FDA及医疗保险和医疗补助服务中心(CMS)数据源整合构建的独特数据集(包含31,000余项提交记录)进行实证研究。相较于FDA现行实践(召回率为10.3%,标准化工作量指标为100%),保守评估显示我们的策略可使召回率改善32.9%,工作量降低40.5%。进一步分析表明,医疗系统因避免置换成本每年可节省约17亿美元,相当于美国全年医疗器械支出的1.1%。本研究结果凸显了采用整体性数据驱动方法改进FDA现行510(k)审批途径的重要价值。