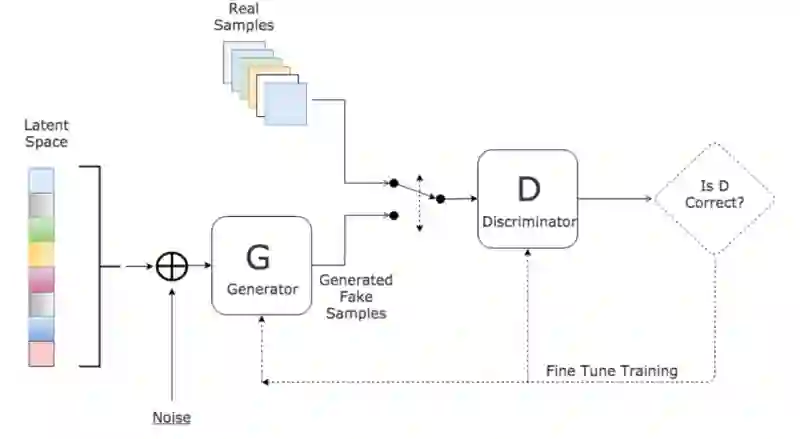
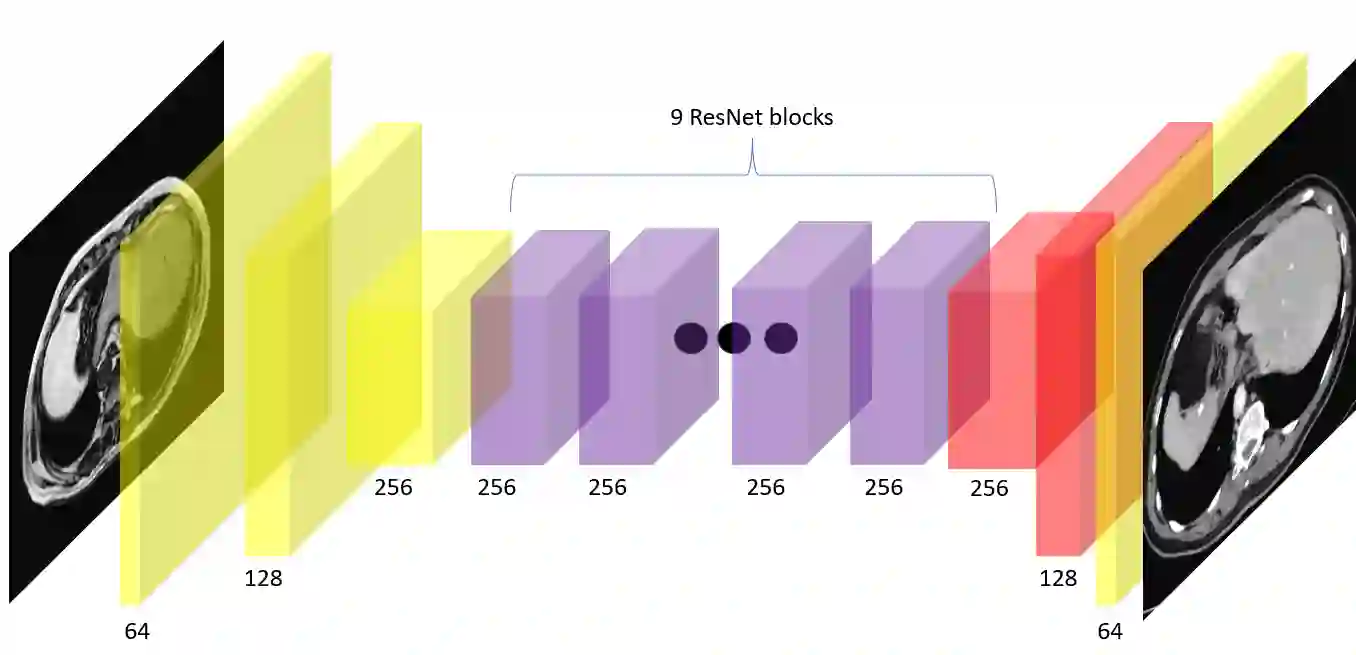
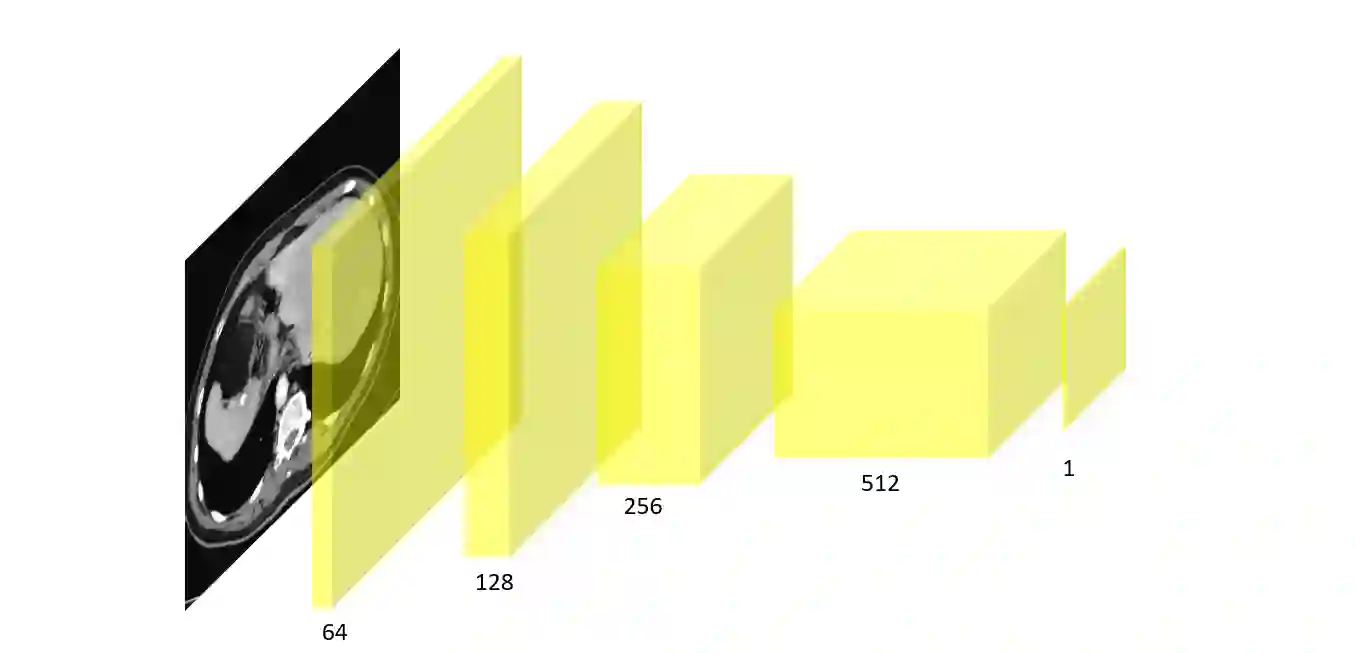
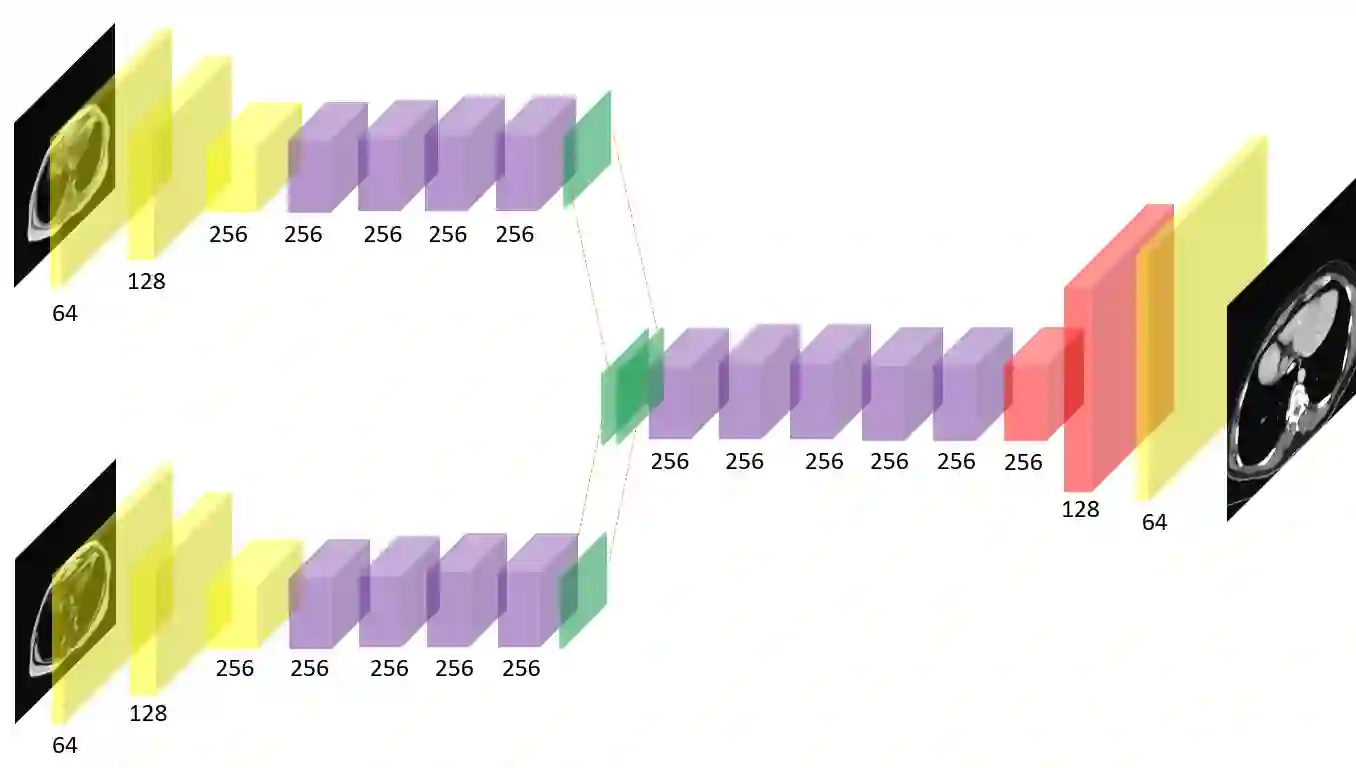
In many clinical settings, the use of both Computed Tomography (CT) and Magnetic Resonance (MRI) is necessary to pursue a thorough understanding of the patient's anatomy and to plan a suitable therapeutical strategy; this is often the case in MRI-based radiotherapy, where CT is always necessary to prepare the dose delivery, as it provides the essential information about the radiation absorption properties of the tissues. Sometimes, MRI is preferred to contour the target volumes. However, this approach is often not the most efficient, as it is more expensive, time-consuming and, most importantly, stressful for the patients. To overcome this issue, in this work, we analyse the capabilities of different configurations of Deep Learning models to generate synthetic CT scans from MRI, leveraging the power of Generative Adversarial Networks (GANs) and, in particular, the CycleGAN architecture, capable of working in an unsupervised manner and without paired images, which were not available. Several CycleGAN models were trained unsupervised to generate CT scans from different MRI modalities with and without contrast agents. To overcome the problem of not having a ground truth, distribution-based metrics were used to assess the model's performance quantitatively, together with a qualitative evaluation where physicians were asked to differentiate between real and synthetic images to understand how realistic the generated images were. The results show how, depending on the input modalities, the models can have very different performances; however, models with the best quantitative results, according to the distribution-based metrics used, can generate very difficult images to distinguish from the real ones, even for physicians, demonstrating the approach's potential.
翻译:在许多临床场景中,需同时使用计算机断层扫描(CT)与磁共振成像(MRI)以全面理解患者解剖结构并规划适宜的治疗策略;这在基于MRI的放射治疗中尤为常见,其中CT始终为剂量投照准备所必需,因其提供了组织辐射吸收特性的关键信息。有时,MRI更适用于靶区勾画。然而,该方法常非最优,因其成本更高、耗时更长,且最重要的是会增加患者压力。为克服此问题,本研究分析了不同配置深度学习模型从MRI生成合成CT扫描的能力,借助生成对抗网络(GANs)——特别是CycleGAN架构——的优势,该架构能够以无监督方式工作且无需配对图像(此类数据本不可得)。多个CycleGAN模型经无监督训练,可从不同MRI模态(包括使用与不使用造影剂)生成CT扫描。针对缺乏真实标注数据的问题,研究采用基于分布的指标定量评估模型性能,并结合定性评估——邀请医师区分真实与合成图像以判断生成图像的真实性。结果表明,根据输入模态的不同,模型性能差异显著;然而,依据所用分布指标获得最佳定量结果的模型,能够生成极难与真实图像区分的合成图像(即使对医师而言亦然),这证明了该方法的潜力。