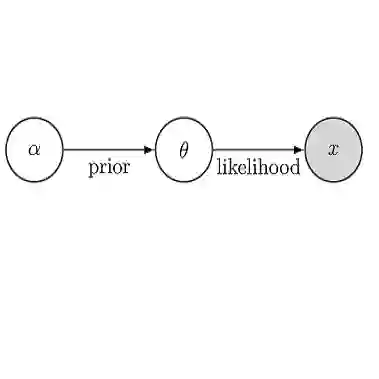
Recent pandemics have highlighted the critical role of infectious disease models in guiding public health decision-making, driving demand for realistic models that can provide timely answers under uncertainty. Compartmental models are widely used to capture disease dynamics, and advances in data availability, computational resources, and epidemiological understanding have allowed the development of models that incorporate detailed representations of population structure, disease progression, and intervention effects. While these improvements improve model fidelity, they also increase model complexity, leading to high-dimensional parameter spaces, intractable likelihoods, and computational challenges for fitting models to limited surveillance data in real time. Existing likelihood-free methods, such as Approximate Bayesian Computation (ABC) and Bayesian Synthetic Likelihood (BSL), have developed largely independently, each with distinct strengths and limitations. We propose an integrated three-stage framework that synthesizes advances from both likelihood-based and likelihood-free method: (1) ABC-based entropy minimization to identify low-dimensional, approximately orthogonal summary statistics; (2) BSL inference using these optimized summaries to construct tractable Gaussian approximations; and (3) Hamiltonian Monte Carlo sampling for efficient posterior exploration. Through SEIR simulation study and application to the 1978 British boarding school influenza outbreak, we demonstrate that our framework achieves reliable parameter estimation and uncertainty quantification while maintaining computational efficiency.
翻译:暂无翻译