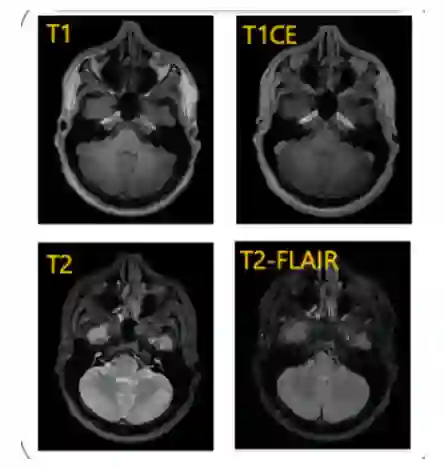
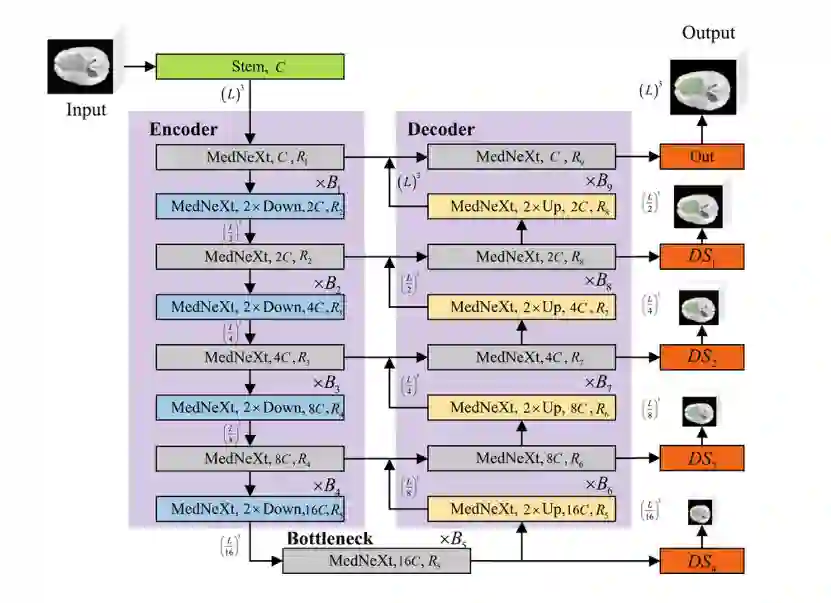
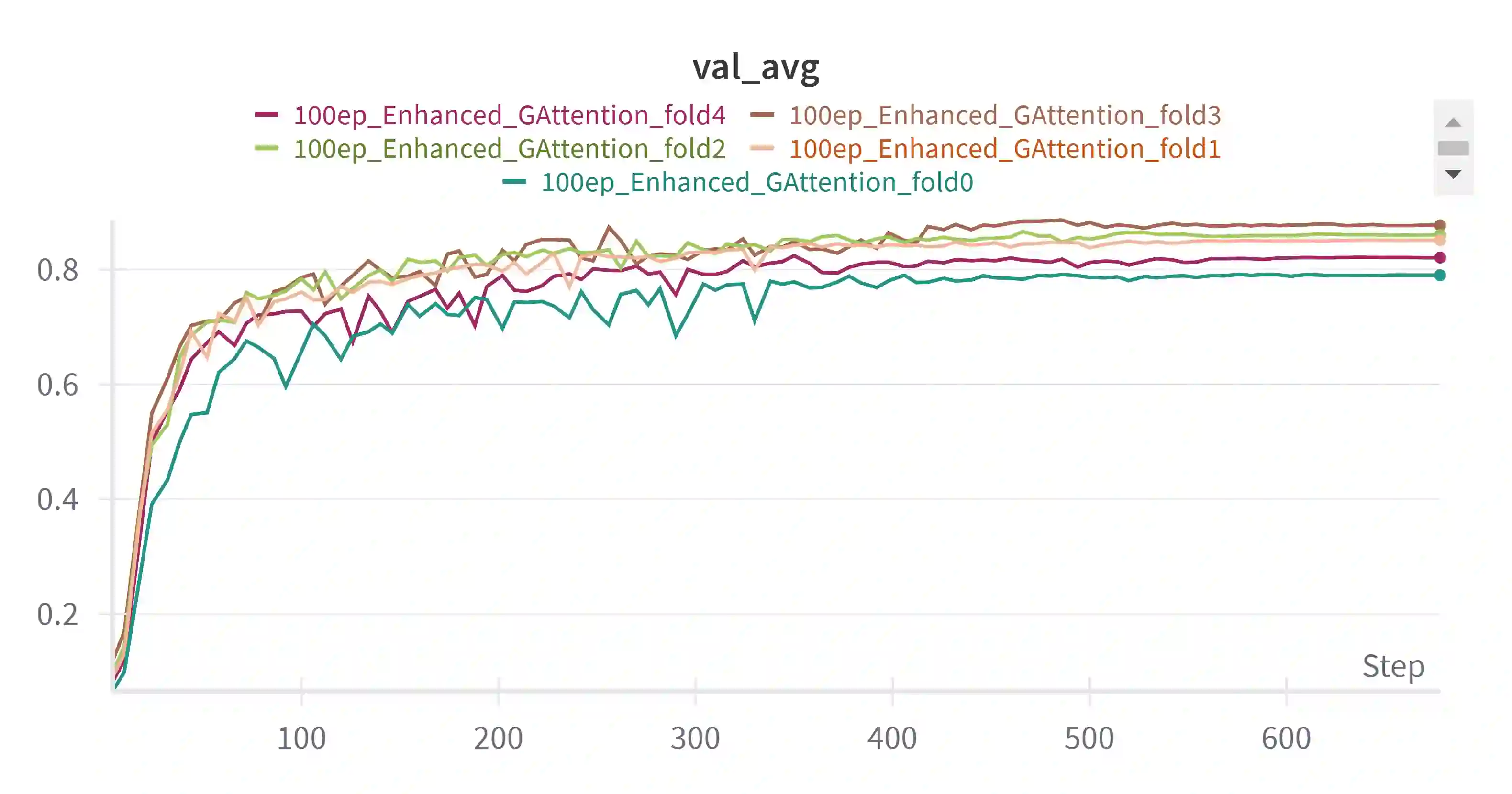
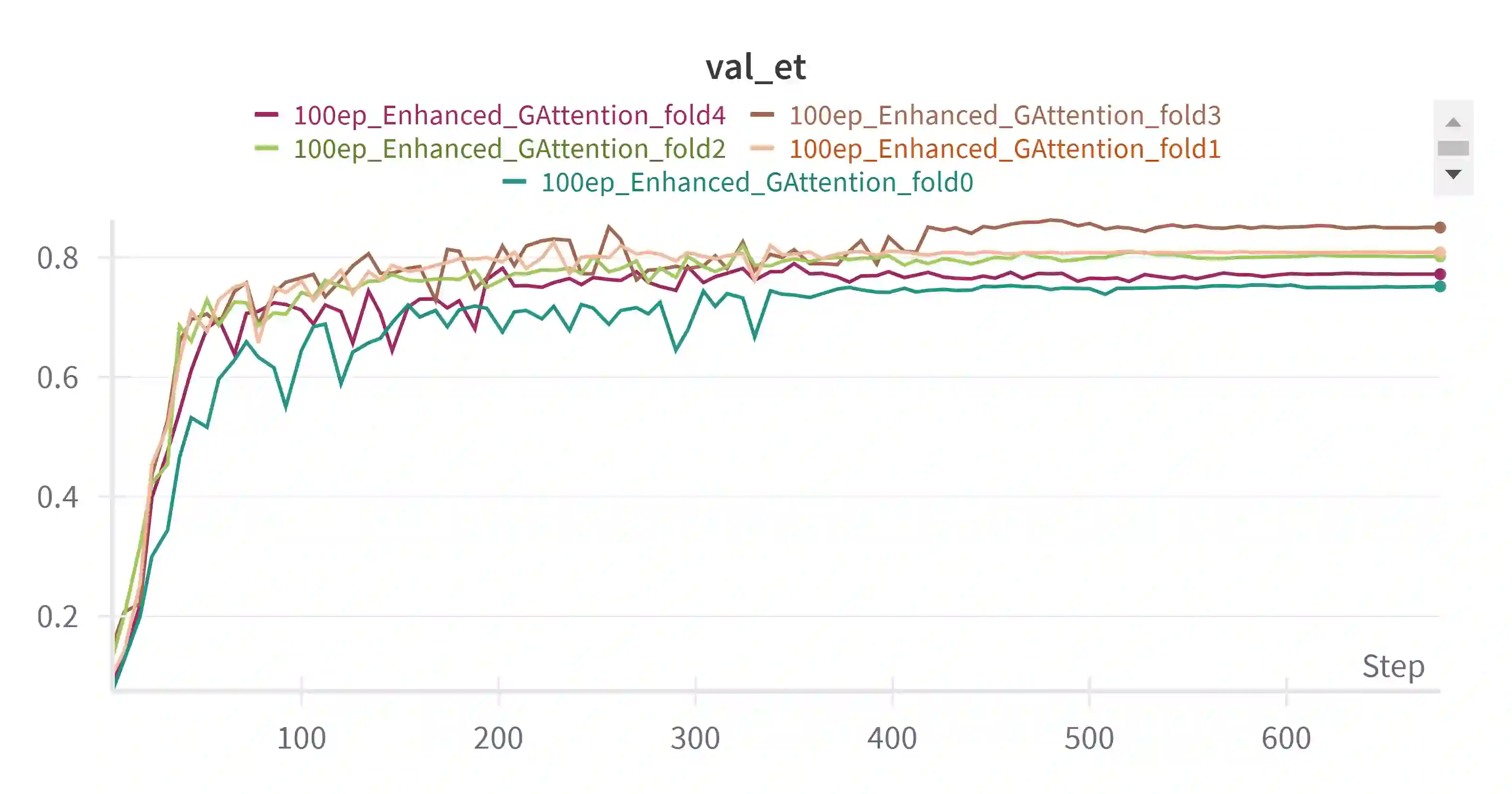
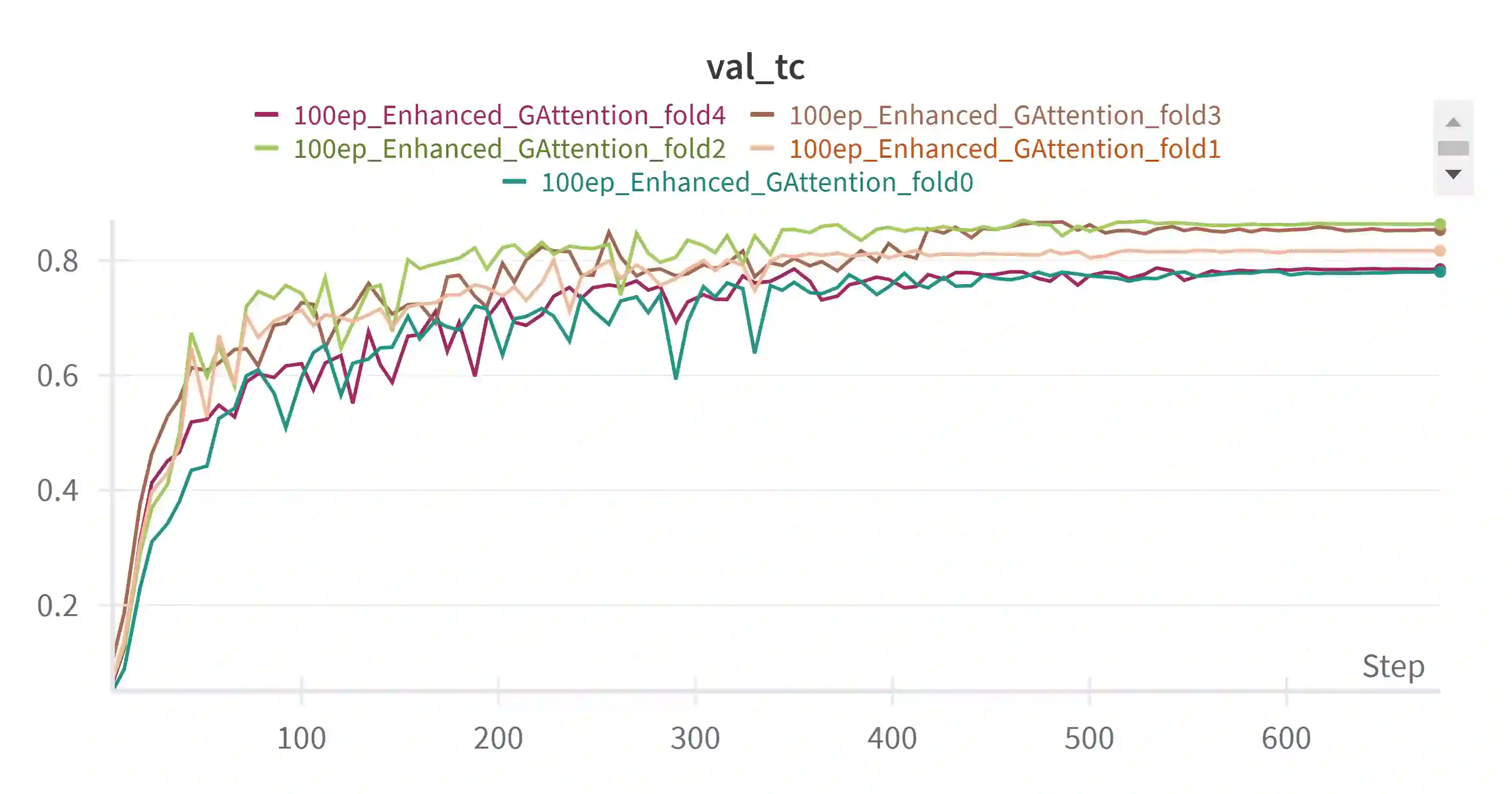
Pediatric brain tumors, particularly gliomas, represent a significant cause of cancer related mortality in children with complex infiltrative growth patterns that complicate treatment. Early, accurate segmentation of these tumors in neuroimaging data is crucial for effective diagnosis and intervention planning. This study presents a novel 3D UNet architecture with a spatial attention mechanism tailored for automated segmentation of pediatric gliomas. Using the BraTS pediatric glioma dataset with multiparametric MRI data, the proposed model captures multi-scale features and selectively attends to tumor relevant regions, enhancing segmentation precision and reducing interference from surrounding tissue. The model's performance is quantitatively evaluated using the Dice similarity coefficient and HD95, demonstrating improved delineation of complex glioma structured. This approach offers a promising advancement in automating pediatric glioma segmentation, with the potential to improve clinical decision making and outcomes.
翻译:儿童脑肿瘤,尤其是胶质瘤,是导致儿童癌症相关死亡的重要原因,其复杂的浸润性生长模式使治疗变得困难。在神经影像数据中对这些肿瘤进行早期、准确的分割对于有效诊断和干预规划至关重要。本研究提出了一种新颖的3D UNet架构,其配备了专门为儿童胶质瘤自动分割定制的空间注意力机制。利用包含多参数MRI数据的BraTS儿童胶质瘤数据集,所提出的模型能够捕获多尺度特征,并选择性地关注肿瘤相关区域,从而提高了分割精度并减少了周围组织的干扰。通过Dice相似系数和HD95对模型性能进行定量评估,结果表明该模型在复杂胶质瘤结构的勾画方面有所改进。该方法为儿童胶质瘤分割的自动化提供了有前景的进展,并有望改善临床决策和预后。